Machines and devices used in modern industry are required to withstand harsh conditions. When the environmental temperature changes, the volume of the materials used to make these devices usually changes slightly, typically by less than 0.01%. Although this may seem like a trivial change, over time this thermal expansion can seriously degrade the performance of industrial systems and equipment.
Materials that contract on heating, or negative thermal expansion materials, are therefore of great interest to industrial engineers. These materials can be mixed with normal materials, which expand on heating, with the final aim of producing a composite material with its thermal expansion adjusted to a particular value, typically zero, and maintained even at the extremely low operating temperatures used in cryogenic and aerospace engineering.
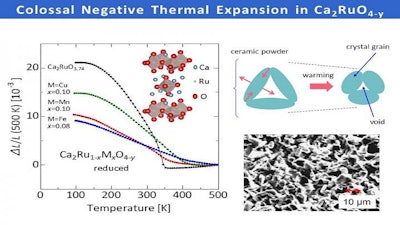
In a new study published in Nature Communications, Nagoya University-led researchers report a reduced ruthenate ceramic material, made up of calcium, ruthenium and oxygen atoms, that shrinks by a record-breaking 6.7 percent when heated. This is more than double the current record for a negative thermal expansion material, and the bulk material expands again when it is cooled.
The results may provide industrial engineers with a new class of composite materials that can be used to increase the accuracy of processes and measurements, to improve the stability of device performance, and to prolong device lifetimes.
The size of the volume change, as well as the operating temperatures for negative thermal expansion can be controlled by changing the composition of the material. When the ruthenium atoms are partially replaced by iron atoms, the temperature window for negative thermal expansion gets much larger. This window extends to above 200 degrees Celsius for the iron-containing material, which makes it particularly promising for industrial use.
Noting that the volume changes were triggered at the same temperature that the reduced ruthenate material changed from a metallic to a non-metallic state, the researchers looked at changes in the arrangement of the atoms using X-ray techniques. They saw dramatic changes on heating, with the internal atomic structure expanding in some directions but contracting in others.
Although the internal structure showed a net contraction, the crystallographic changes were not big enough to explain the giant volume changes in the bulk material. Instead, the researchers turned their attention to the material's overall structure, and found empty voids around the ceramic grains.
"The non-uniform changes in the atomic structure seem to deform the microstructure of the material, which means that the voids collapse and the material shrinks," study corresponding author Koshi Takenaka says. "This is a new way of achieving negative thermal expansion, and it will allow us to develop new materials to compensate for thermal expansion."






















